
Thermit Welding: Operation and Steps [With Set-up Diagram]
Thermit welding is a process where heat generated from an exothermic chemical reaction is used for the fusion. The chemical reaction aluminothermic process occurs between aluminum powder and metal oxide.
This reaction generates a molten metal that acts as filler metal joining the workpieces on solidification. No external source of heat, current, and filler material is utilized in thermit welding.

The process mainly used for joining steel pieces, here thermit comprises iron oxide and aluminum powder. The ratio is 78% iron oxide and 22% aluminum powder. The ratio is decided by the chemical reaction at the burning of aluminum:
8Al + Fe3O4 = 9Fe + 4 Al2O3 + heat (4500˚C, 35 kJ/kg of mixture)
The combustion of iron and aluminum oxide generates heat up to 4500 Degree Fahrenheit. As these two components have a different density, they become separated automatically. The liquid iron fills the ceramic mold built around the welded parts and aluminum oxide slag floats up and removed subsequently.

The thermit process developed by Hans Goldschmidt in the mid-1890s and now used in the repair of steel casting and forging. They design the thermite welding for joining railroad rails, steel pipes, steel wires, a larger cast, and forged components.
Essential Definitions
1. Crucible – It is a vessel where thermite chemical reaction, aluminothermic happens is called a crucible.
2. Mould – The mold is created around the part that requires it to be welded. This mold is destined to receive the molten metal.
3. Mixture – The mixture is the term to describe granules of aluminum and metal oxide. and any alloying metals.
4. Reaction – The aluminothermic process is a chemical reaction between aluminum and metal oxide. The reaction results in super-heated molten material and aluminum oxide slag to be separated.
Railroad Thermite Welding
Thermit Welding Process
It is a chemical welding process where the exothermic reaction is used to provide the needed heat energy. The burning of the thermit, a mixture of aluminum powder and iron oxide in a ratio of 1:3 by weight. generates a temperature of 3000-degree Centigrade.
The preheating of the thermit mixture up to 1300 degrees Centigrade is essential to initiate the reaction.
The thermit mixture reacts as the following chemical reaction:
8Al + Fe3O4 = 9Fe + 4 Al2O3 + heat (4500˚C, 35 kJ/kg of mixture)
A great affinity of aluminum to react with oxygen is the foundation of the thermite chemical reaction. Once reacted with ferric oxide the pure steel settled down while aluminum oxide floats as slag. This all happens due to a big difference in their densities.
Operation and Steps of Thermit Welding
It is essentially a foundry and casting process, where a thermit reaction produces the metal to pour to the obstinate cavity created around the joint. The steps involved in thermit welding are:
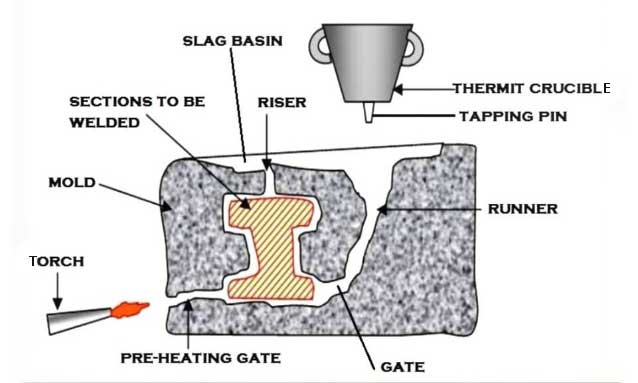
1. It is mandatory to clean and prepare the edges properly the metal parts we will weld.
2. The wax pattern is the need for the joint to be fused. Pour the hot wax to obtain the one.
3. Place the molding box around the joint and the sand is loaded carefully all over the wax pattern. It provides the necessary pouring basin, riser, sprue, and gating system.
4. The bottom opening is used to drain the molten wax which helps to preheat the joint and make it ready for the welding.
5. It is time to mix the thermit in a crucible. This is made from refractory material to withstand the extreme heat and pressure which is produced during this aluminothermic reaction.
6. The igniter like barium oxide or magnesium is left on the head of the mixture and lighted with red hot metal rods.
7. The reaction completes in a very short time, a high heated molten iron flows into the mold cavity which we have created around the joint to be fused.
8. The heated molten material joins the parent metals & solidifies to a homogenous strong joint.
9. The welded joint left as such to cool down slowly.
Copper and Chromium metals have thermit mixtures available as well. They utilize different metal oxides instead of ferrous oxide. Some of the popular thermit reactions with their temperature are mentioned below.
3 CuO + 2 Al → 3Cu + Al2O3 + Heat (4860°C, 270 Kcal)
Cr2O3 + 2Al → 2Cr + Al2O3 + Heat (3000°C, 545 Kcal)
Thermit Welding Steps for Rail
- The rail edges to be welded preparation
- Setting the weld gap
- Apply clamp
- Fix the mold
- Leave the thermit mixture into the crucible
- Preheating of the ends of the rail
- Ignite and let steel flow
- Time to remove the mold
- Remove the excess head metal
- Initiate the grinding
- Final clean grinding
Thermit Welding Process Animation Video
Thermit Welding Kit

A range of welding kits are available as per the need of the welding process.
1. Inch SKV method – The commonest process to join two rails of the train.
2. 1.5-inch SKV method – The process helps to reduce the risk of slag inclusion.
3. 2.75-inch wide gap weld – Used once replacing a thermit weld.
4. Full head repair weld – Fill in once damaged area at the railhead is pulled out.
Uses & Applications

Thermit welding is a very old process of fusion and has been a very useful tool now even.
1. Used to fuse very heavy and thick plates traditionally.
2. It is a preferred choice in joining railroads, thick steel sections, and pipes.
3. Thermit welding does repairing of heavy gears and castings.
4. It is a suitable model to weld large sections such as hull, locomotive rails, and ships.
5. It is useful to weld copper welding cables.
Pros of Thermit Welding
- It is a simple fast method of joining two similar or dissimilar conductive materials.
- It is a very economical process where power supply is not required.
- Thermit welding is possible in distant places where there is no electric supply.
Cons of Thermit Welding
- It is possible to use for ferrous metal parts of bigger sections.
- Light metal and cheap metal fusion are not very economical.
- Welding rate is slow.
- High temperatures may cause a change in the grain structure and distortions of the weld metal.
- Welding may contain gas and slag inclusion.
Frequently Asked Questions
Why only aluminum is used in thermite welding?
The aluminum has a special affinity towards oxygen by its nature. This will react with iron oxide to produce an elemental molten iron. The molten iron gets deposited at the gap to produce a strong and stable joint.
The aluminum on reacting with oxide produces high heat and slag which floats over the molten metal. The aluminothermic reaction generates high heat to melt the elemental iron pool.
Does making thermite illegal?
The thermite used in thermite welding is not illegal as it is not explosive. It becomes illegal if aluminum is supplied in fine mesh, as it is explosive material. Make sure at the time of buying aluminum thermite what the seller is supplying.
Does Thermite Burn Underwater?
A calculated mixture of iron oxide rust and aluminum powder (thermite) needs a high temperature of a hot burning strip of magnesium to ignite it. Once the thermite ignites then it becomes practically impossible to extinguish the fire.
The heat is so high that it can burn through the pavement, engine blocks. The intensity of heat is so high that it stays alive underwater even.
What metals are used for welding railway tracks?
To weld railways rails the thermite is widely used across the world. Copper thermite is the choice for the electric joint using the copper oxide.
Which one is the hottest burning substance known to man?
Thermite seems the man-made hottest burning substance available. The thermite remains a pyrotechnic composition of metal granules and a metal oxide. On chemical reaction, an exothermic oxidation-reduction happens to generate a high degree of heat.
Conclusion
Thermit welding coined in the mid-1890s by Hans Goldschmidt. After a gap of 130 years, it holds a significant place in the modern railways. The railways use continuous weld rail or ribbon rail where rails are joined by flash butt welding to create a track of several kilometers. The union and repair of continuous weld rail is possible only by thermit welding.
Thermit welding is an integral part of modern railways.
Recommendations
Here’s a link to for thermit welding process pdf download
https://en.wikipedia.org/wiki/Exothermic_welding
You Might Also Enjoy.
Understanding The Welding Symbols [Explained With Diagrams]
12 Different Types Of Welding Processes [The Definitive Guide]
